- What is Diabetic Macular Oedema?
- Statistics
- Risk Factors
- Progression
- Symptoms
- Clinical Examination
- How is it Diagnosed
- Prognosis
- Treatment
- Diabetic Macular Oedema Prevention
- References
What is Diabetic Macular Oedema?
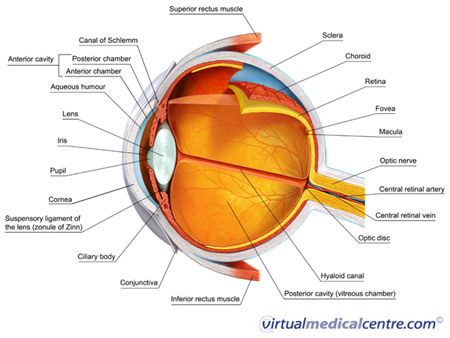
Diabetic macular oedema is a sight-threatening eye condition. It is characterised by the leaking of blood from damaged vessels of the retina (light sensing layer of the eye) into the macula (the section of the eye which enables clear, sharp vision).
 |
For more information, see The Eye and Vision. |
Diabetic macular oedema affects diabetics with proliferative or non-proliferative diabetic retinopathy. The likelihood of developing diabetic macular oedema increases with the progression of non-proliferative diabetic retinopathy and is most likely when the condition progresses to proliferative retinopathy. These conditions are both characterised by reduced blood flow to the retina, to which the eye responds by growing new blood vessels in an attempt to increase blood flow. However the blood vessels are abnormal and weak and may leak into the retina.
Macular oedema may occur and threaten an individual’s sight at any stage of diabetic retinopathy, including in the early stages of non-proliferative disease when there are no visible symptoms. About half of all diabetics with proliferative retinopathy also have macular oedema.
Macular oedema occurs following the breakdown of the blood-retinal barrier due to diabetic retinopathy. The blood-retinal barrier is a layer of cells in the eye which cannot be penetrated by blood and other fluids. It protects the retina by preventing the passing of blood and other fluid into and out of the retina.
Breakdown of the blood-retinal barrier reduces outflow of fluid from the retina. This leads to excessive fluid accumulation in the retina and causes it to swell. The swollen retina affects the macula and may cause decreased central vision (loss of clear sharp vision), vision loss and blindness. Macular oedema is the primary cause of vision loss from diabetic retinopathy.
Macular oedema may be classified as either:
- Diffuse: characterised by generalised leakage from abnormal blood vessels which have grown in the retina. It is often accompanied by capillary loss due to ischaemic maculopathy, another type of diabetic eye disease;
- Focal: characterised by localised areas of leakage in the retina. Leakage occurs primarily from micro aneurysms (microscopic areas of the blood vessel walls which have widened to form sac-like pouches), widening of the capillaries in the retina or other abnormalities of the tiny blood vessels in the eye.
Statistics
Macular oedema affects an estimated 8% of diabetics within 5 years of developing diabetes. The risk of an individual with diabetes developing macular oedema varies depending on diabetes duration, age of onset and patient age. A US study using computer-simulation models estimated that 42% of diabetics will develop macular oedema at some stage in their lifetime. However, there is considerable variation in the results of studies which have investigated the prevalence and incidence of macular oedema.
Risk Factors

There are also differences in the nature of retinopathy depending on diabetes type. Those with Type 1 diabetes are most likely to experience vision loss due to proliferative retinopathy, whereas those with Type 2 diabetes are most likely to experience vision loss as a result of macular oedema. Diabetic maculopathy (which encompasses macular oedema and another condition, macular ischaemia) is more common in type 2, compared to type 1 diabetes.
Progression
Macular oedema may occur as a complication of proliferative or non-proliferative retinopathy; it does not occur in the absence of retinopathy. The condition is characterised by the leaking of fluid from blood vessels in the eye, which have become damaged as a result of retinopathy.
The development of macular oedema appears to be associated with the following changes which occur in diabetic retinopathy:
- Retinal thickening, that is the process of fluid accumulation in the retina which causes it to swell and thicken; and
- Breakdown of the blood-retinal barrier, a layer of the eye which is impenetrable by fluids; it regulates fluid inflow and outflow to the retina.
This leads to extra-cellular fluid accumulation (fluid accumulation in the areas surrounding cells, as opposed to within the cells themselves) in the eye due to breakdown of the blood-retinal barrier. Pressure within the eye increases as the spaces which surround cells in the eye swell with fluid, and subsequently increase pressure in retinal blood vessels.
Changes to the blood-retinal barrier
Non-proliferative diabetic retinopathy is accompanied by changes to the blood-retinal barrier, which are caused by pericyte apoptosis (the death of pericytes) which regulate the growth of blood vessels in the eye. With pericyte apoptosis, new blood vessels in the eye proliferate.
Pericyte apoptosis, and other changes which accompany the unregulated growth of new blood vessels in the eye, affects the blood retinal barrier. The blood retinal barrier is a hydrophobic layer in the eye, meaning it is impenetrable by many fluids including blood. Breakdown of this barrier increasingly allows blood to penetrate and accumulate in the retina.
Changes to the angiotensin-renin system
The angiotensin-renin system refers to interactions between the enzymes angiotensin and renin which regulate the amount of fluid in the body as well as blood pressure. Enzymes and chemicals involved in the angiotensin-renin system (renin, angiotensinogen and angiotensin-converting enzyme (ACE), angiotensin II and prorenin) are widely expressed in the eyes and are involved in many ocular processes.
Angiotensin affects cell growth and blood pressure, and increases the permeability of blood vessels. Increased concentrations of prorenin, ACE and angiotensin II in the blood or the eyes are associated with the development of diabetic retinopathy.
Concentrations of angiotensin in the eye also appear to influence the development of macular oedema. Increases in angiotensin II concentrations occur alongside increases in concentrations of vascular endothelial growth factors (VEGF) in the eye. VEGF is the hormone which promotes blood vessel growth, such as the growth of tiny, abnormal blood vessels in the eyes in retinopathy.
Progression of symptoms
Vision loss from macular oedema can occur at any stage of retinopathy, even in the early stages of asymptomatic, non-proliferative disease.
Symptoms
Diabetic retinopathy often has no obvious symptoms until major bleeding causing vision loss occurs. Macular oedema can occur as a complication of either non-proliferative or proliferative diabetic retinopathy. Symptoms include:
- Blurred vision;
- Wavy central vision; and/or
- Altered colour perception.
Diabetics should undergo regular screening to detect changes before vision symptoms occur.
Clinical Examination

- A visual acuity test which is a test of how well you can see. Visual acuity may be tested using a Snellen Chart (a chart containing lines of letters which progressively decrease in size and from which you are asks to read to test your vision);
- Examination of the eyes for signs of vessel damage by slit lamp biomicroscopy, that is; using a special lamp and a low powered microscope. This enables the examiner to view microscopic changes which are not visible to the naked eye. Your pupils must be dilated using eye drops before this test.
How is it Diagnosed
Diabetic macular oedema occurs as a complication of diabetic retinopathy. Only diabetics affected by retinopathy will develop macular oedema. Diabetic retinopathy is detected through slit lamp biomicroscopy of the retina. If the doctor finds evidence of diabetic retinopathy, slit lamp biomicroscopy will also be used to examine the macular, looking for abnormal vessel growth in close proximity to the macula. If vascular abnormalities near the macula are identified, the doctor will need to conduct further tests to characterise macular oedema.
Fluorescein angiogram
Fluorescein angiogram refers to a procedure in which fluorescent dye is injected into a vein before an x-ray is taken. The dye circulates in the blood vessels and creates a contrast on the x-ray. This makes it easier to identify irregularities in the blood vessel system, such as where blood vessels are leaking.
If slit lamp biomicroscopy produces evidence of macular oedema, a fluorescein angiogram may be used to identify leakage points and other abnormalities of the eye’s blood vessels, or the cause of an unexplained loss in visual acuity.
Optical coherence tomography
Optical coherence tomography is a non-invasive technique which uses patterns of light scattering in the eye to generate images of the inside of the eye. The thickness and shape of internal structures can be determined by the pattern of light scattering.
Optical coherence tomography may be used to assess macula and retina thickness during diagnosis, and/or to assess changes in macular oedema throughout treatment.
Other tools
Other measures which may be used in the diagnosis of diabetic retinopathy include:
- Heidelberg retinal photography may be used to assess retinal thickness in order to quantify the extent of macular oedema;
- Retinal thickness analysis may be used to quantify the extent of macular oedema;
- Tonometry may be used to measure pressure within the eye. Pressure is increased in macular oedema due to the accumulation of fluid in the eye.
Prognosis
Untreated, diabetic macular oedema is a sight threatening condition. It is more likely to progress and cause vision loss in diabetics with sub-optimal diabetes control. For example; those who are unable to control their blood sugar, blood pressure and cholesterol within the recommended ranges by modifying their lifestyle and using medications.
Several treatments for macular oedema reduce the likelihood of vision loss by approximately 50%, however repeat treatment is often needed. Some diabetics with macular oedema experience vision loss in spite of aggressive treatment.
Treatment
Adequate control of blood sugar, blood pressure and cholesterol minimise the chances of diabetic retinopathy progressing and/or causing macular oedema. If you are diagnosed with retinopathy your doctor may recommend medication and lifestyle changes to improve your diabetes control.
In diabetics with retinopathy, the ideal blood sugar HbA1c levels (a measure of blood glucose levels) is around 7%. Anti-hypertensives (medicines which control blood pressure, or BP) may be prescribed with the aim of lowering systolic BP to <130 mm HG. Fibrates and statins may be prescribed to control cholesterol levels.
Treatments for all types of diabetic retinopathy can cure vision loss symptoms and prevent further vision loss. However, it is important to note that they do not cure the underlying vessel damage which causes the symptoms and are unlikely to restore vision which has already been lost.
Macular laser surgery
Macular oedema is typically treated with focal laser surgery. The procedure involves directing several hundred laser beams into the eye near the affected area of macula in a grid pattern. The treatment is usually completed in a single session, however if both eyes are affected by macular oedema, it is normal to treat one eye at a time.
Focal laser surgery slows leaking from damaged blood vessels and reduces the amount of fluid which enters the macula. It reduces the likelihood of vision loss by 50%, although repeat surgery is sometimes necessary. There are also a number of adverse side effects associated with the treatment, including:
- Decreased central vision (clear sharp vision) following treatment;
- Partial vision loss, or loss of vision in a defined area of the visual field, due to a laser burn directed in the wrong area of the eye;
- Fibrous growth behind the retina;
- Neovascularisation (growth of tiny abnormal blood vessels in the choroid, the membrane which separates the retina from the sclera (the covering of the eyeball).
If you have proliferative retinopathy as well as macular oedema, you will undergo macula laser surgery before the commencement of retinal laser surgery. This minimises the risk of macular oedema worsening as a result of retinal laser surgery.
Vitrectomy
Vitrectomy is a surgical procedure which involves removing blood from the eye. In theory it should be an effective treatment for macular oedema which has not improved with laser treatment. However, research results have shown that vitrectomy is often ineffective for treating macular oedema. It may, however, be used to treat diabetics with severe proliferative retinopathy characterised by bleeding in the retina or vitreous fluid (a gel like layer which protects the retina). It may be particularly useful for diabetics with severe proliferative retinopathy and macular oedema.
Intravitreal steroid therapy
Intravitreal steroid therapy involves the injection of steroids such as cortisol into the eye. It may be used to treat diabetics with macular oedema which persists after treatment with laser surgery, as well as those with extensive hard deposits in the macula. The hard deposits form from the blood and fluids which leak from blood vessels in the retina.
It may also be used as adjunct therapy (a secondary therapy which improves the effectiveness of the primary therapy) in combination with retinal laser surgery. The treatment reduces leakage from blood vessels and inhibits the growth of fibrous tissues and granulomas (clusters of tissue). However, research shows that intravitreal steroid injection achieves better short vision outcomes compared to laser surgery. Conversely, long term outcomes are better in diabetics treated with laser surgery.
Intravitreal steroid therapy improves visual acuity by up to 50% and reduces future vision loss. However, repeat injections are usually required and serious side effects including an increased risk of several sight-threatening conditions are associated with this treatment. Your doctor will discuss these risks with you before you undergo treatment.
Intravitreal anti-vascular endothelial growth factor injections
Research investigating the effectiveness of intravitreal injections has reported better outcomes compared to laser surgery. For example, in one study half the diabetics treated with intravitreal anti-VEGF experienced visual improvements compared to 28% of those treated with laser surgery. Other studies have also reported improved visual acuity with intravitreal anti-VEGF injections, and that this treatment makes visual deterioration less likely. However, treatment involves an average of nine intravitreal injections annually, as well as monthly checkups. One intravitreal anti-VEGF medication, ranibizumab (Lucentis), was recently added to the pharmaceutical benefits scheme meaning that eligible individuals receive a subsidy to cover the cost of treatment.
Intravenous monoclonal anti-tumour necrosis factor
Monoclonal anti-tumour necrosis factor (infliximab, marketed as Remicade) is a medicine used in the treatment of other eye conditions and inflammatory arthritis and Crohn’s disease. It is emerging as a treatment for diabetic macular oedema, however to date only one small research study has been conducted to assess its effectiveness. Further large research studies are needed to determine the risks and benefits of monoclonal anti-tumour necrosis factors as therapy for macular oedema.
Review
Following treatment for diabetic macular oedema, you will need to return to your doctor after 3 to 4 months so that the doctor can re-assess your eyes and determine whether or not the treatment was effective and if you need further treatment. Be sure to keep any follow-up doctor’s appointments.
Support services for patients with severe visual deterioration
If your visual acuity deteriorates significantly, you may be able to register as legally blind or partially sighted. This may assist you to access appropriate support services. If your treatment fails to relieve your vision symptoms, you may need to access other support services to help you adjust to your new level of vision, for example rehabilitation and social services. You may also wish to speak to a counsellor if you find it difficult to cope with your condition. Speak to your doctor about referral to counsellors and other support services.
Diabetic Macular Oedema Prevention

Optimal management of diabetes is also an important component of prevention for diabetics with proliferative retinopathy and/or macular oedema. Research shows that optimal diabetes control reduces the risk of macular oedema in diabetic patients by 26% over 10 years.
Screening for changes in blood flow to the retina is the key preventative measure, however at least 22% of Australian diabetics are not screened as often as recommended by Australian guidelines, which recommend screening at least every 2 years. Currently there is little evidence about why diabetics fail to have their eyes screened according to recommendations. Cost of screening was identified as a barrier in one US study, while a UK study found that underestimating the severity of disease was a key factor in screening non-compliance in that country.
All diabetics should have comprehensive eye examinations, at least every 2 years. If proliferative retinopathy or macular oedema is diagnosed, prompt referral (within 4 weeks) to a specialist who can treat the condition is essential. If your doctor gives you a referral letter for a specialist appointment, be sure that you attend the appointment.
References
- Mitchell P, Foran S. Guidelines for the management of diabetic retinopathy [online]. Canberra, ACT: National Health and Medical Research Council; 2008 [cited 28 February 2011]. Available from: URL link
- Facts about diabetic retinopathy [online]. Bethesda, MD: National Eye Institute; October 2009 [cited 22 February 2011]. Available from: URL link
- Spurling G, Askew D, Jackson C. Retinopathy: Screening recommendations. Aust Fam Physician. 2009;38(10):780-3. [Abstract | Full text]
- Ockrim Z, Yorston D. Managing diabetic retinopathy. BMJ. 2010;341:c5400. [Abstract]
- Diabetic retinopathy [online]. San Francisco, CA: American Academy of Ophthalmology; 2008 [cited 28 February 2011]. Available from: URL link
- Williams R, Airey M, Baxter H, et al. Epidemiology of diabetic retinopathy and macular oedema: A systematic review. Eye (Lond). 2004;18(10):963-83. [Abstract | Full text]
- Watkins PJ. Retinopathy. BMJ. 2003;326(7395):924-6. [Abstract | Full text]
- Stewart MW. Chapter 1: Pathophysiology of diabetic retinopathy. In: Browning DJ (ed). Diabetic Retinopathy: Evidence-based management. New York: Springer; 2010: 1-31. [Book]
- Macular edema symptoms [online]. San Francisco, CA: American Academy of Ophthalmology (AAO); 2011 [cited 5 August 2011]. Available from: URL link
- Diabetic retinopathy: Initial and follow-up evaluation [online]. San Francisco, CA: International Council of Ophthalmology; 3 January 2011 [cited 22 February 2011]. Available from: URL link
- Viswanath K, McGavin DD.Diabetic retinopathy: Clinical findings and management. Community Eye Health. 2003;16(46):21-4. [Full text]
- Sfikakis PP, Markomichelakis N, Theodossiadis GP, et al. Regression of sight-threatening macular edema in type 2 diabetes following treatment with the anti-tumor necrosis factor monoclonal antibody infliximab. Diabetes Care. 2005;28(2):445-7. [Abstract | Full text]
- Product Information: Lucentis. North Ryde, NSW: Novartis Pharmaceuticals Pty Ltd; 25 October 2011.
All content and media on the HealthEngine Blog is created and published online for informational purposes only. It is not intended to be a substitute for professional medical advice and should not be relied on as health or personal advice. Always seek the guidance of your doctor or other qualified health professional with any questions you may have regarding your health or a medical condition. Never disregard the advice of a medical professional, or delay in seeking it because of something you have read on this Website. If you think you may have a medical emergency, call your doctor, go to the nearest hospital emergency department, or call the emergency services immediately.