- What is Diabetic Eye Disease: Non-Proliferative Diabetic Retinopathy?
- Statistics
- Risk Factors
- Progression
- Symptoms
- Clinical Examination
- How is it Diagnosed
- Prognosis
- Treatment
- Diabetic Eye Disease: Non-Proliferative Diabetic Retinopathy Prevention
- References
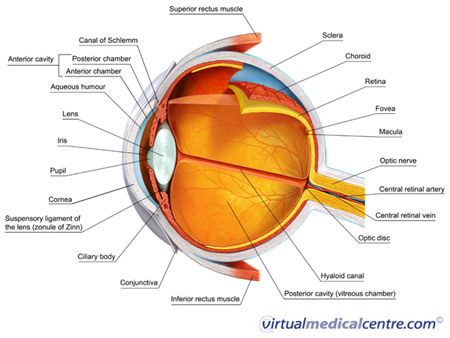
What is Diabetic Eye Disease: Non-Proliferative Diabetic Retinopathy?
Non-proliferative diabetic retinopathy, previously called background retinopathy, is the earliest stage of diabetic eye disease. Microscopic changes occur in the blood vessels of the eye in non-proliferative disease; however, the changes typically do not produce symptoms and are not visible to the naked eye. Non-proliferative disease progresses from mild to moderate to severe.
Non-proliferative diabetic retinopathy is initially characterised by microaneurysms (microscopic blood-filled bulges in the artery walls) which may burst and leak into the retina. Tiny spots or dots of blood may accumulate in the retina, but they usually do not produce noticeable symptoms in the early stages of the disease. As the disease progresses, hard exudates (accumulations of fluid that has leaked from blood vessels), abnormalities in the growth of microscopic blood vessels in the retina, and bleeding from the veins that feed the retina may occur.
While non-proliferative diabetic retinopathy is not itself a sight-threatening condition, it can trigger macular oedema or macular ischaemia, which are other forms of diabetic retinopathy that may cause rapid vision loss at any stage of non-proliferative disease. In addition, the vascular changes that occur in non-proliferative retinopathy lead to retinal ischaemia (lack of blood flow to the retina) and trigger progression to sight-threatening proliferative disease. As the severity of non-proliferative retinopathy increases, the risk of developing sight-threatening proliferative diabetic retinopathy also increases.
Proliferative diabetic retinopathy
Proliferative diabetic retinopathy is characterised by neovascularisation – that is, the growth of abnormal new blood vessels in the retina. The vessels are weak and may burst and bleed into the retina or vitreous fluid (fluid surrounding the retina), causing vision loss.
Diabetic macular oedema
At all stages of non-proliferative diabetic retinopathy, there is also a risk of macular oedema, a sight-threatening condition that occurs as a consequence of the vascular changes of diabetic retinopathy. Macular oedema is the most common cause of diabetes-associated vision loss. It occurs when abnormal new blood vessels burst and bleed into the macula (the area of the eye that enables clear, sharp vision), disrupting vision.
Ischaemic maculopathy
Ischaemic maculopathy is an untreatable form of diabetic retinopathy characterised by the loss of capillaries (tiny blood vessels) that provide blood flow to the macula.
Statistics

Each year, an estimated 4.5% of the diabetic population are diagnosed with diabetic retinopathy for the first time, although a larger proportion of some diabetic groups, particularly the elderly and Indigenous Australians, will develop the condition each year. In a 5-year period, 11% of Australian diabetics aged ? 40 years will develop retinopathy. However, the likelihood of retinopathy varies depending on the length of time a person has had diabetes, their age when they developed diabetes, and their current age. A 5-year incidence of 22% was reported in the Blue Mountains Eye Study, in which all participants were over 50.
The prevalence of retinopathy amongst diabetics increases with progression of diabetes and duration since diabetes onset. At the time of diagnosis, 6% of diabetics have some evidence of retinopathy. After 10 years of diabetes, 7% of patients have evidence of retinopathy, and after 25 years, some 90% will be affected by retinopathy of varying degrees. Evidence of retinopathy is almost universal amongst type 1 diabetics after 2 years duration, and amongst type 2 diabetics 30 years after onset.
Importantly, prevalence and severity of diabetic retinopathy appears to be declining. The decline is attributed to better diabetes management and treatment, which enables diabetics to better control their blood pressure and other metabolic abnormalities.
Globally, non-proliferative diabetic retinopathy was estimated to affect 171 million people in 2000. Global prevalence is expected to increase to 366 million by 2030.
Risk Factors
Diabetes is the key risk factor for non-proliferative diabetic retinopathy, and all individuals with type 1 or type 2 diabetes mellitus are at risk of developing the condition. Some individuals with abnormal blood sugar levels who do not develop diabetes are also at risk of diabetic retinopathy. In these groups of individuals, risk factors for development and progression of the disease include:
- Duration of diabetes: The primary risk factor. Risk increases with increasing diabetes duration;
- Early diabetes onset: Diagnosis before puberty and diagnosis before age 30 are associated with increased risk of retinopathy;
- Type 1 diabetes: More likely to cause retinopathy compared to type 2 diabetes;
- Poor glycaemic control: The risk of diabetic retinopathy increases in line with the extent of blood sugar abnormalities;
- Hypertension or high blood pressure;
- High cholesterol;
- Pregnancy: Associated with an increased risk of developing non-proliferative retinopathy. Some studies have shown more rapid progression of existing disease in pregnancy;
- Kidney impairment: Asociated with diabetic retinopathy, though it appears that diabetic retinopathy leads to kidney impairment rather than vice versa;
- Genetic susceptibility: However, the genetics of diabetic retinopathy are not well understood; and
- Cataract surgery.
Early diabetes diagnosis
Early diabetes diagnosis (before age 30) is associated with a higher risk of diabetic retinopathy. Amongst patients who received a diabetes diagnosis before age 30, proliferative retinopathy affects 25% after 15 years duration, and 55% after 20 years (compared to 20% of individuals diagnosed after age 30 with ? 20 years disease duration).
Diabetes
In patients with type 2 diabetes, available evidence suggests 40% have some type of retinopathy, with severe, sight-threatening retinopathy affecting 8.2% of the type 2 diabetic population. Data regarding prevalence of non-proliferative diabetic retinopathy in type 1 diabetes is more limited, though one study reported 82% prevalence for any type or severity of retinopathy and 32% prevalence for sight-threatening retinopathy.
This study and others indicate a higher prevalence of retinopathy in type 1 diabetes compared to type 2 diabetes. However, more recent research has reported higher risk in those with type 2 diabetes. This conflicting result is thought to be because of the changing epidemiology of diabetic retinopathy. Type 1 diabetics now have an increased relative risk of the condition compared to type 2 diabetics, due to improvements in glycaemic control in type 1 diabetes patients.1
Poor glucose control
The more an individual’s blood sugar levels differ from normal, the more likely they are to develop retinopathy. Individuals with impaired glucose tolerance (such as in metabolic syndrome) who do not develop diabetes also have a risk of diabetic retinopathy, and 7.9% of those who do not develop diabetes nonetheless show signs of diabetic eye disease.
Some data show associations between improved glycaemic control (control of blood sugar levels) and reduced progression of retinopathic changes. There is also evidence that optimal glycaemic control significantly reduces development and progression of retinopathic changes by up to 54% in type 1 diabetic patients and up to 25% in type 2 diabetic patients, in comparison to conventional treatment. However, there is also conflicting evidence in relation to this. Other studies that have assessed the effect of tight glycaemic control on the progression of retinopathy have reported no benefit of tight compared to standard glycaemic control.
Hypertension
Hypertension is a risk factor for changes in blood flow to the eye, such as those that characterise retinopathy, and a number of other eye diseases. Hypertension is associated with eye diseases even in the absence of co-existing diabetes. Hypertension also influences the risk of non-proliferative diabetic retinopathy. High-quality evidence suggests that treating hypertensive diabetics with blood pressure reducing therapies decreases the risk of diabetic retinopathy.
Progression

Increased oxidative stress is associated with both hyperglycaemia and diabetes. Oxidative stress also appears to play a role in the development of diabetic retinopathy. Each of the pathways through which diabetic retinopathy is thought to occur are associated with the production of free radical molecules and increased oxidative stress in the retina.
Those which are linked to the development of non-proliferative diabetic retinopathy include:
- The sorbitol and aldose reductase pathway by which glucose is metabolised;
- Changes to retinal blood flow;
- Changes to the blood vessels; and
- Changes to the blood–retinal barrier (the barrier that normally prevents blood from entering the retina).
Sorbitol and aldose reductase pathways
The sorbitol–aldose reductase pathway regulates the metabolism of glucose, and is a key pathway in the development of non-proliferative diabetic retinopathy. Aldose reductase is an enzyme that catalyses the breakdown of glucose to form sorbitol. In individuals with high blood sugar, this process leads to an accumulation of sorbitol, which may cause:
- Damage to vascular cell walls; and
- Damage to and apoptosis (death) of retinal pericytes. Retinal pericytes play an important role in regulating the growth of blood vessels in the eye. In particular, they prevent the proliferation of vascular cells. Damage to retinal pericytes therefore enables vascular cells to proliferate.
Pericyte apoptosis is a mechanism in the development of the vascular changes that characterise non-proliferative retinopathy, and also influences neovascularisation in proliferative retinopathy. In non-proliferative disease, the loss of pericytes leads to the formation of microaneurysms (blood-filled bulges) in the venous capillaries due to weak capillary walls. New blood vessel cells proliferate around the microaneurysms in an attempt to strengthen the vessel. However, blood vessel growth is unregulated and may eventually cause venous occlusion (closing of the vein) and ischaemia (lack of blood flow). Ischaemia triggers the growth of new blood vessels in an attempt to increase blood flow, and is the key mechanism by which the neovascularisation characteristic of proliferative retinopathy occurs.
Changes in the sorbitol–aldose reductase pathway also lead to the development of precursors to advanced glycation end-products (discussed below), and may increase oxidative stress which is associated with the development of retinopathy.
Retinal blood flow changes
Retinal blood flow increases in non-proliferative diabetic retinopathy due to contraction of blood vessels that feed the retina. This causes retinal ischaemia (lack of blood flow to the retina), because although more blood flows into the eye, contraction of blood vessels means that not enough blood reaches the retina. Retinal blood flow changes appear to play a role in the development of non-proliferative retinopathy. In the later stages of disease when larger retinal blood vessels widen to counter ischaemia, blood flow decreases.
Changes to the blood–retinal barrier
Non-proliferative diabetic retinopathy is accompanied by changes to the blood–retinal barrier that result from the loss of pericytes (caused by sorbitol accumulation, as discussed above). The blood–retinal barrier is a layer through which most fluids cannot penetrate. The purpose of the barrier is to protect the retina. When it breaks down, fluid (including blood) is allowed to penetrate the retina.
Vascular changes
The widening of retinal veins and arterioles (small veins) in diabetic retinopathy are characteristic of vascular changes that occur throughout the body in the early stages of diabetes.
Other mechanisms that influence the development of non-proliferative diabetic retinopathy
A number of other mechanisms are thought to influence the development of non-proliferative diabetic retinopathy, including:
- Pregnancy: Can lead to inflammation and damage to the blood vessel system, as a result of the woman’s changing hormone profile during pregnancy; and
- Increased levels of homocysteine, an amino acid associated with cardiovascular disorders.
Progression of symptoms
Non-proliferative diabetic retinopathy is characterised by the growth of microaneurysms that eventually cause retinal ischaemia. When blood flow to the retina is insufficient, neovascularisation, or the abnormal growth of microscopic blood vessels, occurs in the retina. Neovascularisation is characteristic of the proliferative or more advanced stage of the disease, and can affect vision.
In non-proliferative retinopathy, vision is not affected unless macular oedema occurs at the same time. The condition may also cause vision loss if neovascularisation occurs and it progresses to proliferative retinopathy. In severe cases of non-proliferative disease, progression to proliferative disease typically occurs within 15 months.
Symptoms

It is therefore important for diabetics to have their eyes examined regularly, so that retinopathic changes can be identified in the early stages, and steps take to prevent irreversible vision loss. Treatments for diabetic retinopathy can help prevent future vision loss, but they are not effective in restoring vision that is already lost.
Diabetes is the key risk factor for retinopathy. Duration of diabetes and sub-optimal blood sugar and blood pressure control make retinopathy more likely, so a doctor will likely ask about your diabetes history when assessing the likelihood of retinopathy and your need for an eye examination. In particular, it’s important to give your doctor information about the duration of your diabetes, your past and current blood sugar levels, and the medication you use to treat diabetes. You should also tell the doctor about any health conditions you have experienced such as obesity, hypertension, kidney problems, and any previous eye conditions or problems.
As pregnancy increases the likelihood of retinopathy, diabetic women who are planning to conceive should discuss diabetes management with their doctor in their preconception care consultations.
Clinical Examination
Clinical assessment to screen for diabetic retinopathy should involve:
- A visual acuity test (for example by using a Snellen chart, which has rows of letters of decreasing size. The person being tested reads through the lines to enable the doctor to assess the quality of their vision);
- Examination of the eyes for signs of vessel damage. This is done by dilating the pupils and using a slit lamp. Eye drops are usually used to dilate the pupils.
A special lamp called a slit lamp is used to examine the retina when screening for diabetic retinopathy. Usually the person’s pupils are dilated using eye drops before the examination, and a microscope is used in order to detect changes that are not visible to the naked eye. You will probably be referred to an ophthalmologist or optometrist for the examination, though some general practitioners may conduct eye exams.
Note that if you have an eye condition called closed angle glaucoma, you should not have your pupils dilated. Tell your doctor if you have previously been diagnosed with closed angle glaucoma.
Screening for diabetes in patients with features of diabetic retinopathy
Diabetes is undiagnosed in up to one third of cases. Undiagnosed diabetics may notice visual problems before they notice other symptoms of diabetes. In these cases, diabetes may be diagnosed as a result of the eye examination. If you find out you have diabetes as a result of an eye examination that shows retinopathy, it is very important to visit a general practitioner for assessment and management of your diabetes.
How is it Diagnosed
A slit lamp examination of the eyes is usually sufficient to diagnose diabetic retinopathy. There are a number of other ways the condition can be diagnosed if a slit lamp examination is not available, or if the diagnosis remains questionable after this examination. These include:
- Photographic examination: Photos of different sections of the eye are taken and sent to a specialist for examination. Digital or standard photos may be used; and
- Fluorescein angiogram: A special dye is injected into the blood vessels. The dye flows through the blood vessels and, due to its contrasting colour, a doctor is able to identify the pattern of blood flow, including, for example, sections of the retinal vessels in which blood flow is obstructed.
Investigations to determine diabetes status and control
In some instances, the assessment of diabetic retinopathy may also involve assessment of your diabetes control (e.g. your blood sugar levels). As such, the doctor may draw blood for laboratory assessment or measure your blood pressure.
Prognosis

Non-proliferative disease may also cause macular oedema, even in the very early stages. Macular oedema is the most common cause of visual loss in diabetic retinopathy.
Treatment
Non-proliferative disease is asymptomatic, and treatment aims to slow progression to proliferative sight-threatening disease and reduce the risk of macular oedema occurring as a complication. Adequate control of blood sugar, blood pressure and cholesterol minimise the chances of disease progression and are the key measures taken to manage individuals with diabetic retinopathy. Adequate control is typically achieved using dietary interventions and medications. For example, high blood pressure may be managed using beta blockers or angiotensin-converting enzyme inhibitors.
More Information
 |
For more information on diabetes, including different types of diabetes, blood glucose and dietary control, and long-term complications, as well as some useful videos, see Diabetes. |
Diabetic Eye Disease: Non-Proliferative Diabetic Retinopathy Prevention
Prevention of diabetic retinopathy involves screening for early stage disease through eye examinations and diabetes management.
Eye examination
While retinopathy cannot be prevented, progression of the disease and vision loss can be stopped in patients with non-proliferative retinopathy through frequent eye examination and timely treatment when proliferative retinopathy or macular oedema is diagnosed. This is the key preventative measure. Preventative screening and timely treatment where appropriate has been shown to be highly cost-effective and reduces the risk of vision loss from retinopathy by 95%. However, because a considerable portion of diabetics either fail to screen for and/or treat their condition, the screening approach is not as effective as it could be.
At least 22% of Australian diabetics are not screened as often as recommended by the National Health and Medical Research Council’s (NHMRC) recommendations (bi-annually). Your doctor may therefore ask you about the last time you had an eye examination and encourage you to have your eyes screened every two years, in line with the NHMRC’s recommendations.
The NHMRC recommend eye examinations be performed at the time of diabetes diagnosis and at least bi-yearly thereafter. While a considerable proportion of type 2 diabetics (> 25%) have evidence of retinopathy at the time of diagnosis, it is unusual for type 1 diabetics to develop retinopathy in the 5 years following diabetes onset.
In other countries, annual screening is recommended. This also provides extra opportunities for diabetics to discuss their condition with a health professional, and increases the opportunities for identification of other eye conditions, which are particularly likely to affect older individuals. It has been suggested that more regular screening may increase screening compliance, as people would be less likely to forget when their next appointment is due if it occurs every year; however, some question the cost-effectiveness of annual screening in the non-proliferative stage of disease, when the risk of progression to proliferative diabetic retinopathy is generally low.
In Australia, annual screening is only recommended for diabetics with a high risk of disease progression and vision loss. These individuals include:
- Indigenous Australians;
- Australians from a non-English speaking background;
- Those who have had diabetes for a longer period of time, as risk of retinopathy and vision loss increases with increasing duration of diabetes; or
- Those with poorly controlled blood sugar, blood pressure or cholesterol.
Those who have previously been diagnosed with non-proliferative diabetic retinopathy should also have more regular eye examinations, either yearly, half-yearly or quarterly, depending on the severity of the disease.
Children who were diagnosed with diabetes before puberty should have their eyes re-examined at the onset of puberty. Diabetic women who become pregnant should also have their eyes assessed when the pregnancy is diagnosed. If there is evidence of retinopathy, these women need regular eye examinations throughout pregnancy. Monitoring is not required in cases of gestational diabetes, unless diabetes persists post-pregnancy.
In addition to regular screening for asymptomatic disease, is it important to see a doctor and have your eyes examined if you experience any of the following symptoms:
- Reduced night vision;
- Blurred vision;
- Vision blocked by patches or streaks; or
- Sudden vision loss.
Diabetes management

Diabetics and individuals with poor glycaemic control (e.g. as in metabolic syndrome) should ensure they are educated about the role of obesity and metabolic abnormalities in exacerbating diabetic retinopathy, as well as the importance of adhering to a diabetic diet in prevention of the disease. Limiting alcohol consumption is also important. While alcohol consumption alone is not associated with diabetic retinopathy, the consumption of empty calories from alcohol makes adhering to a calorie-restricted diabetic diet very difficult.
References
- Guidelines for the management of diabetic retinopathy [online]. Canberra, ACT: National Health and Medical Research Council; 2008 [cited 28 February 2011]. Available from: URL link
- Spurling G, Askew D, Jackson C. Retinopathy: Screening recommendations. Aust Fam Physician. 2009;38(10):780-3. [Abstract | Full text]
- Ockrim Z, Yorston D. Managing diabetic retinopathy. BMJ. 2010;341:c5400. [Abstract]
- Watkins PJ. Retinopathy. BMJ. 2003;326(7395):924-6. [Abstract | Full text]
- Fong DS, Aiello L, Gardner T, et al. Diabetic retinopathy. Diabetes Care. 2003;26(Suppl 1):S99-102. [Abstract | Full text]
- Diabetic retinopathy: Symptoms [online]. London: National Health Service; 16 December 2009 [cited 16 June 2011]. Availbale from: URL link
- Diabetic retinopathy [online]. San Francisco, CA: American Academy of Ophthalmology; 2008 [cited 28 February 2011]. Available from: URL link
- Trad MJ. Diabetic retinopathy. In: Onofrey BE, Skorin L, Holdeman NR. Ocular Therapeutics Handbook: A clinical manual (2nd edition). Philadelphia, PA: Lippincott Williams & Wilkins; 2005: 520-6. [Book]
- Stewart MW. Chapter 1: Pathophysiology of diabetic retinopathy. In: Browning DJ (ed). Diabetic Retinopathy: Evidence-based management. New York: Springer; 2010: 1-31. [Book]
- Chapter 9: Management of proliferative diabetic retinopathy. In: Wu G. Diabetic Retinopathy: The essentials. Philadelphia, PA: Lippincott Williams & Wilkins; 2010: 122-37. [Book]
- Facts about diabetic retinopathy [online]. Bethesda, MD: National Eye Institute; October 2009 [cited 22 February 2011]. Available from: URL link
- Diabetic retinopathy: Initial and follow-up evaluation [online]. San Francisco, CA: International Council of Ophthalmology; 3 January 2011 [cited 22 February 2011]. Available from: URL link
All content and media on the HealthEngine Blog is created and published online for informational purposes only. It is not intended to be a substitute for professional medical advice and should not be relied on as health or personal advice. Always seek the guidance of your doctor or other qualified health professional with any questions you may have regarding your health or a medical condition. Never disregard the advice of a medical professional, or delay in seeking it because of something you have read on this Website. If you think you may have a medical emergency, call your doctor, go to the nearest hospital emergency department, or call the emergency services immediately.