- What is Pre-Invasive Ductal Carcinoma (Ductal Carcinoma In Situ; DCIS; Breast Cancer)
- Statistics on Pre-Invasive Ductal Carcinoma (Ductal Carcinoma In Situ; DCIS; Breast Cancer)
- Risk Factors for Pre-Invasive Ductal Carcinoma (Ductal Carcinoma In Situ; DCIS; Breast Cancer)
- Progression of Pre-Invasive Ductal Carcinoma (Ductal Carcinoma In Situ; DCIS; Breast Cancer)
- Symptoms of Pre-Invasive Ductal Carcinoma (Ductal Carcinoma In Situ; DCIS; Breast Cancer)
- Clinical Examination of Pre-Invasive Ductal Carcinoma (Ductal Carcinoma In Situ; DCIS; Breast Cancer)
- How is Pre-Invasive Ductal Carcinoma (Ductal Carcinoma In Situ; DCIS; Breast Cancer) Diagnosed?
- Prognosis of Pre-Invasive Ductal Carcinoma (Ductal Carcinoma In Situ; DCIS; Breast Cancer)
- How is Pre-Invasive Ductal Carcinoma (Ductal Carcinoma In Situ; DCIS; Breast Cancer) Treated?
- Pre-Invasive Ductal Carcinoma (Ductal Carcinoma In Situ; DCIS; Breast Cancer) References
What is Pre-Invasive Ductal Carcinoma (Ductal Carcinoma In Situ; DCIS; Breast Cancer)
| Ductal carcinoma is a type of breast carcinoma (cancer), making up approximately 80% of all cases of breast cancer diagnosed in Australia. Ductal carcinoma may be divided into in-situ carcinoma (DCIS) and invasive ductal carcinoma (IDC). Non-invasive cancers stay within the milk ducts or lobules of the breast. They do not invade normal tissues unlike invasive breast cancers.Ductal carcinoma in situ is sometimes called a pre-malignant condition. It describes an abnormal growth of the milk duct cells. The cells share similar microscopic features with invasive breast cancer, but are confined to the milk ducts (see the image below). The term ‘in situ’ means that the cancer has not yet penetrated through the basement membrane (the membrane at the base of the epithelial lining of ducts or glands). |
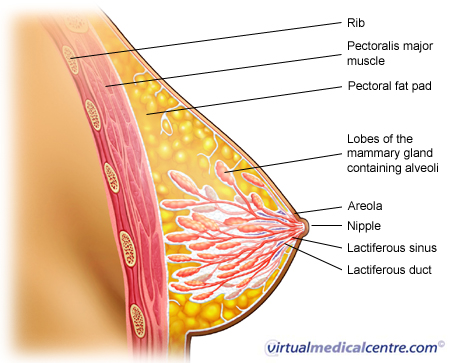
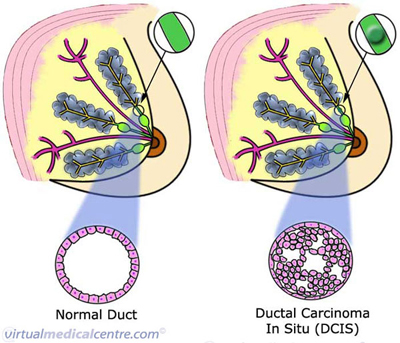
Statistics on Pre-Invasive Ductal Carcinoma (Ductal Carcinoma In Situ; DCIS; Breast Cancer)
Pre-invasive ductal carcinoma in-situ was diagnosed in about 1,724 Australian women in 2008, and increase from 998 in 1997. The age standardised diagnosis of incident ductal carcinoma in situ increased from 11 per 100,000 in 1997 to 15 per 100,000 in 2008.4
Current evidence suggests that invasive ductal carcinoma starts as ductal carcinoma in-situ. Ductal carcinoma is the most common histological type of invasive breast cancers. In 2008 there were 10,527 invasive ductal carcinoma diagnoses.4
Risk Factors for Pre-Invasive Ductal Carcinoma (Ductal Carcinoma In Situ; DCIS; Breast Cancer)

- Female gender;
- Increasing age;
- A family history of breast cancer;
- Previous history of DCIS;
- Breast diseases such as complex fibroadenoma (non-cancerous tumour of the breast), atypical hyperplasia (accumulation of abnormal cells in a breast duct), sclerosing adenosis (excessive growth of tissues in the breast lobules) and intraductal papilloma (non-cancerous tumour of the breast duct);
- Hormonal factors: Women who begin menstruation early, women who have experience late menopause, late or few pregnancies, or who are obese. Some evidence is emerging that long term oestrogen therapy (either oral contraceptive pill or hormone replacement therapy (HRT)) may increase risk, as may a diet which is high in fat;
- Others: Exposure to radiation (e.g. from x-rays or CT); inherited gene abnormalities such as BRCA1 and BRCA2 mutations.
Progression of Pre-Invasive Ductal Carcinoma (Ductal Carcinoma In Situ; DCIS; Breast Cancer)
The term DCIS describes an abnormal growth of cancerous cells in the milk ducts of the breast. The abnormal cells have similar features to those of invasive breast cancer, but are not able to spread beyond the milk duct into surrounding normal breast tissue.
It is estimated that women who have had DCIS are 4–12 times more likely to develop subsequent invasive breast cancer despite adequate treatment. No reliable factors have been identified which predispose people with DCIS to subsequently develop invasive breast cancer but the risk may be higher when the DCIS lesion displays certain microscopic features.
Symptoms of Pre-Invasive Ductal Carcinoma (Ductal Carcinoma In Situ; DCIS; Breast Cancer)
DCIS is usually detected through mammography (x-rays of the breast). Rarely, it may present as a lump which can be felt or as an incidental finding on tissue sampling of breast tissue. Changes in the skin of the breast and/or a bloody nipple discharge may also sometimes be present.
How is Pre-Invasive Ductal Carcinoma (Ductal Carcinoma In Situ; DCIS; Breast Cancer) Diagnosed?
Initial tests
Any breast symptom, such as a lump or nipple discharge, is assessed with the ‘triple test’. This includes examination of the breast, imaging of the breast through mammography (breast x-ray) or ultrasound, and sampling of the breast tissue with fine needle aspiration (FNA), core biopsy or open biopsy.
Further tests
Following a diagnosis of breast cancer, blood tests including full blood count and liver function tests may be used to assess the possibility of the cancer spreading to the liver or bone marrow. Other imaging tests, including chest x-ray, bone scan, positron emission tomography (PET) scan, abdominal CT or liver ultrasound may also be used if symptoms suggest that the cancer has spread.
Prognosis of Pre-Invasive Ductal Carcinoma (Ductal Carcinoma In Situ; DCIS; Breast Cancer)

Women with DCIS are at increased risk of developing invasive breast cancer in the future. However, as most cases of DCIS are now treated, it is not known exactly how high this risk is, or how quickly the progression from in situ to invasive cancer is likely to occur.
Local recurrence (the tumour regrowing in the same area) rates of DCIS depend on the type of treatment that is implemented. There is a 1.4% recurrence rate when DCIS is treated with both conservative breast surgery and radiotherapy.
How is Pre-Invasive Ductal Carcinoma (Ductal Carcinoma In Situ; DCIS; Breast Cancer) Treated?
The goal of therapy for DCIS is to prevent the development of invasive breast cancer and typically includes localised treatment (surgery and radiotherapy), chemotherapy and hormonal therapy.
Surgical treatment
Surgery aims to achieve total disease control through removal of the primary breast tumour. Surgical treatment can be divided into two major streams: Breast conserving surgery with complete local excision (CLE) and axillary dissection, or total mastectomy with axillary dissection (removal of axillary lymph nodes).These two procedures have been shown to confer similar survival rates at 5 and 10 years post-procedure. The decision on choice of treatment is therefore based on clinical features, such as the size and location of the tumour:
- Larger, widespread cancers or cancer near the nipple may require mastectomy (removal of all breast tissue). This can be followed by reconstructive surgery;
- Smaller tumours or tumours located in the outer quadrant in larger breasts may be removed by lumpectomy (removal of lump) or partial mastectomy (removal of selected areas of breast tissue). Radiotherapy usually follows the procedure;
- In selected cases, with small well-differentiated tumours, omission of axillary dissection may be considered.
Radiotherapy
Radiation therapy after surgical treatment has been proven to reduce the risk of disease recurrence and development of invasive breast cancer. Studies have shown that at 15 years, the rate of developing invasive breast cancer was about half in people who had both radiation therapy and surgery as compared to those who only had surgical therapy.
Systemic therapy
Chemotherapy has never been investigated or used in the treatment of women with DCIS. Only tamoxifen (a drug that blocks the oestrogen receptor) which is an adjuvant (a treatment given after another treatment) hormonal treatment has been investigated in studies to determine its effectiveness in the treatment of DCIS. Data from studies suggest that tamoxifen may reduce the risk of subsequent local invasive breast cancer in women who have had surgical treatment for DCIS which is discussed further in the next section.
Hormonal therapy
Women with tumours that are proven on histology to express oestrogen receptors should be offered hormone therapies such as tamoxifen or ovarian ablation therapy (procedure whereby ovaries are intentionally damaged to decrease production of oestrogen). Tamoxifen may be considered for people treated with breast-conserving therapy and radiation therapy, especially for those with DCIS expressing oestrogen receptors. The benefit of tamoxifen in reducing risk of subsequent invasive breast cancer has not been investigated for DCIS not expressing oestrogen receptors.
More information
 |
For more information on breast cancer, types of breast cancer and its investigations and treatments, as well as some useful videos, see Breast Cancer. |
Pre-Invasive Ductal Carcinoma (Ductal Carcinoma In Situ; DCIS; Breast Cancer) References
- Australian Institute of Health and Welfare & National Breast Cancer Centre 2006. Breast cancer in Australia: an overview, 2006. Cancer series no. 34. cat. no. CAN 29. Canberra: AIHW. [cited 28 September 2015]. Available from: [URL Link]
- Braunwald, Fauci, Kasper, Hauser, Longo, Jameson. Harrison’s Principles of Internal Medicine. 16th Edition. 2001. McGraw-Hill. [Book]
- Cotran RS, Kumar V, Collins T. Robbins. Pathological Basis of Disease. Sixth Ed. WB Saunders Company 1999. [Book]
- Australian Institute of Health and Welfare. Breast Cancer in Australia: an Overview 2012. Cancer series No. 71. Cat. No. CAN67. 2012. [cited 28 September 2015]. Available from: [URL Link]
- Kumar P, Clark M. Clinical Medicine. Fourth Edition. WB Saunders, 1998. [Book]
- National Breast Cancer Centre. The clinical management of ductal carcinoma in situ, lobular carcinoma in situ and atypical hyperplasia of the breast First Edition. National [cited 28 September 2015] Available from: [URL Link]
- National Health and Medical Research Council. Clinical practice guidelines for the management of early breast cancer. 2001. Second edition. National Breast Cancer Centre [cited 28 September 2015] Available from: [URL Link]
- Schwartz, GF, Solin, LJ, Olivotto, IA, et al. Consensus Conference on the Treatment of In Situ Ductal Carcinoma of the Breast, April 22-25, 1999. Cancer 2000; 88:946. [Abstract]
- Talley NJ, O’Connor Simon. Clinical Examination. Fourth Edition. 2001. MacLennan & Petty. [Book]
- Jemal A, Siegel R, Xu J et al. Cancer statistics 2010. CA Cancer J Clin. 2010; 60: 277. [Abstract | Full Text]
- Cancer Australia. The investigation of a new breast symptom. 2006 [cited 28 September 2015]: Available from: [URL link]
- Goodwin A, Parker S, Ghersi D et al. Post-operative radiotherapy for ductal carcinoma in situ of the breast- a systematic review of the randomised trials. Breast. 2009; 18: 143. [Abstract]
- Petrelli F, Barni S. Tamoxifen added to radiotherapy and surgery for the treatment of ductal carcinoma in situ of the breast: A meta-analysis of 2 randomised trials. Radiother Oncol. 2011. 100; 195-9. [Abstract]
- Harris JR, Lippman E, Morrow M. Diseases of the Breast. Lippincott Williams and Wilkins. 2009. [Book]
- Boyages J, Delaney G, Taylor R. Predictors of local recurrence after treatment of ductal carcinoma in situ: a meta-analysis. Cancer. 1999; 85: 616. [Abstract]
- Degnim AC, Visscher DW, Berman HK, et al. Stratification of breast cancer risk in women with atypia: a Mayo cohort study. J Clin Oncol. 2007; 25: 2671. [Abstract | Full text]
- National Comprehensive Cancer Network. NCCN Guidelines Version 2/2012 Breast Cancer. Available from: [URL Link]
All content and media on the HealthEngine Blog is created and published online for informational purposes only. It is not intended to be a substitute for professional medical advice and should not be relied on as health or personal advice. Always seek the guidance of your doctor or other qualified health professional with any questions you may have regarding your health or a medical condition. Never disregard the advice of a medical professional, or delay in seeking it because of something you have read on this Website. If you think you may have a medical emergency, call your doctor, go to the nearest hospital emergency department, or call the emergency services immediately.







