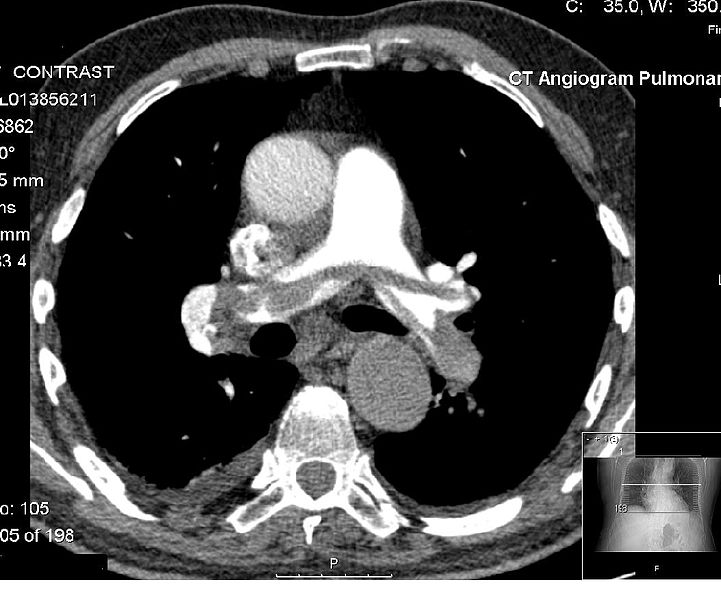
PI-88 is a new experimental drug that inhibits tumour growth by reducing the formation of new blood vessels into tumours. Docetaxel is a standard second-line treatment offered to patients with non-small-cell lung cancer who havenât responded to first-line therapies (platinum-based drugs or radiotherapy). Of this group of patients, only 20% remain progression-free 6 months after starting docetaxel treatment. The PR88202 study has been designed to compare two different cancer treatments (docetaxel only, and docetaxel plus PI-88) and to work out which is more effective against the cancer. It is hoped that the combination of PI-88 with docetaxel will allow patients to extend the time it takes for their disease to progress, and also to improve their quality of life.
Official Title
Trial of PI-88 with Docetaxel in Advanced Non-Small-Cell Lung Cancer (NSCLC)
Conditions
Carcinoma, Non-Small-Cell Lung
Study Type
Interventional
Study Design
Treatment, Randomized, Open Label, Historical Control, Parallel Assignment, Safety/Efficacy Study
Further Details
PR88202 is an open-label randomized study. In the initial phase of the study, patients will be randomized to receive weekly docetaxel alone, or PI-88 in combination with weekly docetaxel. Both groups will receive docetaxel (30 mg/m2), administered by intravenous infusion on days 1, 8 and 15 of a 28-day cycle. The second group only will receive PI-88 (250 mg/day) in addition to docetaxel; PI-88 will be administered by subcutaneous injection on days 1-4, 8-11 and 15-18 of each cycle. The primary efficacy endpoint is the non-progression rate at 6 months. In the extension phase of the study, patients in the combination arm who have stable disease or an objective response after up to six treatment cycles will remain on PI-88 alone as maintenance therapy. Patients who initially receive docetaxel alone and who have disease progression or unacceptable toxicity before the completion of six cycles will be eligible to receive PI-88 alone as third-line therapy.
Study Start
February 2004
Eligibility & Criteria
Ages Eligible for Study: 18 Years and above, Genders Eligible for Study: Both Criteria:Inclusion Criteria:- histologically or cytologically confirmed stage IIIb or IV NSCLC that has progressed during or after first-line treatment – measurable disease by spiral CT chest scan, as defined in RECIST criteria – performance status 0-1 (ECOG) – life expectancy at least 2 months – adequate hemopoietic, renal and hepatic function Exclusion Criteria:- current symptomatic central nervous system (CNS) involvement – prior or co-existent malignancies – significant non-malignant disease – acute or chronic gastrointestinal (GI) bleeding in last two years – inflammatory bowel disease – abnormal bleeding tendency – patients at risk of bleeding due to open wounds or planned surgery – clinically significant hemoptysis within the past 4 weeks – bilirubin > upper limit of normal (ULN) – ALT and AST>2.5 times ULN, or > 1.5 times ULN if alkaline phosphatase>2.5 times ULN – alkaline phosphatase > 5 times ULN, unless patient has bone metastases – myocardial infarction, stroke or congestive heart failure within last 3 months – prior treatment with docetaxel – concomitant treatment with aspirin (>100 mg/day), NSAIDs (except selective COX-2 inhibitors, warfarin (>1 mg/day), heparin, LMWH, anti-platelet drugs, CYP3A4 inhibitors – women who are pregnant or breast-feeding – women of child-bearing potential not using adequate contraception – history of allergy and/or hypersensitivity to anti-coagulants or thrombolytic agents, especially heparin – history of immune-mediated thrombocytopenia, thrombotic thrombocytopenic purpura or other platelet disease – allergy to polysorbate 80 (component of Taxotere®) – uncontrolled or serious infection in last 4 weeks
Total Enrolment
100
Contact Details
Brian Creese +61 7 3273 9175 brian.creese@progen.com.auKathy Mackenzie +61 2 3273 9192 kathy.mackenzie@progen.com.auAustralia, Australian Capital TerritoryCanberra Hospital, Canberra, Australian Capital Territory, 2605, Australia; Not yet recruiting Desmond Yip, MD +61 2 6244 2220 dyip@med.usyd.edu.au Jenny May +61 2 6244 3688 Jenny.May@act.gov.au Desmond Yip, MD, Principal InvestigatorAustralia, New South WalesRoyal North Shore Hospital, St. Leonards, New South Wales, 2065, Australia; Recruiting Nick Pavlakis, MD +61 2 9926 5020 pavlakis@med.usyd.edu.au Sally McCowatt +61 2 9926 5049 smccowat@doh.health.nsw.gov.au Nick Pavlakis, MD, Principal InvestigatorSydney Cancer Centre, Royal Prince Alfred Hospital, Camperdown, New South Wales, 2050, Australia; Recruiting Michael Boyer, MD +61 2 9515 5494 boyer@bigpond.com Annabel Childs +61 2 9515 7028 Annabel.Childs@email.cs.nsw.gov.au Michael Boyer, MD, Principal InvestigatorPrince of Wales Hospital, Randwick, New South Wales, 2031, Australia; Recruiting Craig Lewis, MD +61 2 9382 2606 LewisC@sesahs.nsw.gov.au Clare Bligh +61 2 9382 2466 BlighC@sesahs.nsw.gov.au Craig Lewis, MD, Principal InvestigatorNewcastle Mater Misericordiae Hospital, Waratah, New South Wales, 2298, Australia; Recruiting Fiona Abell, MD +61 2 4921 1148 fiona.abell@mater.health.nsw.gov.au Ian Howitt +61 2 4921 1868 Ian.Howitt@newcastle.edu.au Fiona Abell, MD, Principal InvestigatorSydney Haematology and Oncology Clinics, Hornsby, New South Wales, 2077, Australia; Recruiting Gavin Marx, MD +61 2 9476 5844 gmarx@shoc.com.au Jane Garrad +61 2 9476 5844 jgarrad@shoc.com.au Gavin Marx, MD, Principal InvestigatorConcord Repatriation Hospital, Concord, New South Wales, 2139, Australia; Recruiting Michael Boyer, MD +61 2 9476 5844 boyer@bigpond.com Lili Truong +61 2 9767 5112 lili.truong@email.cs.nsw.gov.au Michael Boyer, MD, Principal InvestigatorAustralia, QueenslandMater Hospital, South Brisbane, Queensland, 4101, Australia; Recruiting Paul Mainwaring, MD +61 7 3840 6166 Paul_Mainwaring@mater.or.au Ken Musgrave +61 7 3840 8697 Ken_Musgrave@mater.org.au Paul Mainwaring, MD, Principal InvestigatorPrince Charles Hospital, Chermside, Queensland, 4032, Australia; Recruiting Rick Abraham, MD +61 2 3350 8520 rcabraham@bigpond.com Jo-Ann Horn +61 7 3212 5863 Joanne_Horn@health.qld.gov.au Rick Abraham, MD, Principal InvestigatorPrincess Alexandra Hospital, Woolloongabba, Queensland, 4102, Australia; Not yet recruiting Keith Horwood, MD +61 2 3240 2584 keith@qldoncology.com.au Margaret Dauth +61 2 3240 2584 respmed@health.qld.gov.au Keith Horwood, MD, Principal InvestigatorNambour General Hospital, Nambour, Queensland, 4560, Australia; Not yet recruiting Geoff Hawson, MD +61 418 870 140 geoffrey@hawson.org Chris Cocks +61 7 5470 6202 christine_cocks@health.gld.gov.au Geoffrey Hawson, MD, Principal InvestigatorAustralia, South AustraliaThe Queen Elizabeth Hospital, Woodville, South Australia, 5011, Australia; Recruiting Ken Pittman, MD +61 8 8222 6148 Ken’Pittman@nwahs.sa.gov.au Greer Dymott +61 8 8222 8719 Greer.Dymmott@nwahs.sa.gov.au Ken Pittman, MD, Principal InvestigatorAustralia, VictoriaThe Alfred Hospital, Prahran, Victoria, 3187, Australia; Recruiting Jeremy Shapiro, MD +61 3 9509 1866 Jeremy.Shapiro@med.monash.edu.au Sarah Bullen +61 3 9276 2629 S.Bullen@alfred.org.au Jeremy Shapiro, MD, Principal Investigator Border Medical Oncology, Wodonga, Victoria, 3690, Australia; Recruiting Craig Underhill, MD +61 2 6055 3200 CUnderhill@bordermedonc.com.au Chris Hodgkins +61 2 6056 7542 CHodgkins@bordermedonc.com.au Craig Underhill, MD, Principal InvestigatorAustralia, Western AustraliaSir Charles Gairdner Hospital, Nedlands, Western Australia, 6009, Australia; Recruiting Michael Millward, MD +61 8 9346 3823 millward@cyllene.uwa.edu.au Bonnie McLeod +61 8 9346 4438 bonnie.mcleod@health.wa.gov.au Michael Millward, MD, Principal Investigator
All content and media on the HealthEngine Blog is created and published online for informational purposes only. It is not intended to be a substitute for professional medical advice and should not be relied on as health or personal advice. Always seek the guidance of your doctor or other qualified health professional with any questions you may have regarding your health or a medical condition. Never disregard the advice of a medical professional, or delay in seeking it because of something you have read on this Website. If you think you may have a medical emergency, call your doctor, go to the nearest hospital emergency department, or call the emergency services immediately.