- What is Molar Pregnancy (Gestational Trophoblastic Disease, GTD)?
- Statistics
- Risk Factors
- Progression
- Symptoms
- Clinical Examination
- How is it Diagnosed
- Prognosis
- Treatment
- References
What is Molar Pregnancy (Gestational Trophoblastic Disease, GTD)?
 |
|
Image courtesy of Blausen Medical Communications. |
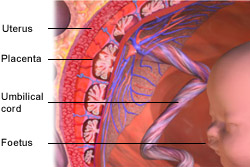
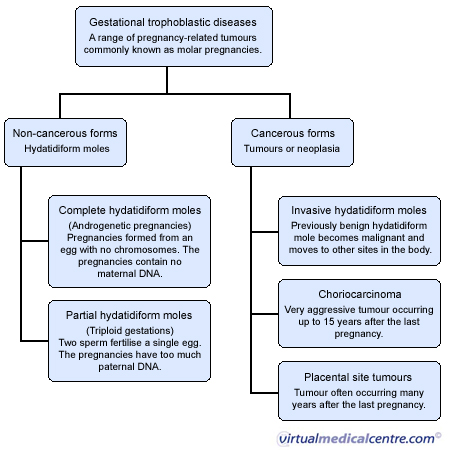
Gestational trophoblastic disease (GTD), also known as molar pregnancy, is a range of pregnancy related tumours. Molar pregnancies occur when the placenta (or afterbirth) of a defective pregnancy transforms into a tumour. It can occur following normal or abnormal fertilisation. The tumours are unique because they arise from placental tissues and therefore contain maternal and paternal genes (whereas most tumours arise only from the tissues of the individual in which they are found).They can be either benign (i.e. non-cancerous) or malignant (i.e. cancerous). Benign forms of GTD are known as hydatidiform moles, whereas cancerous moles are known as gestational trophoblastic tumours, or gestational trophoblastic neoplasia.
Benign GTD tumours are either complete or partial hydatidiform moles. They represent incomplete pregnancies in which the placenta grows but the embryo (i.e. the foetus) does not. In a normal pregnancy, a single chromosome from each of the mother (egg) and father (sperm) join to form an embryo which is surrounded by the placenta. It develops into a foetus and is then born as a baby. In molar pregnancies this process of fertilisation is defective.
A complete hydatidiform mole is an androgenetic pregnancy (i.e. a pregnancy which contains only paternal and non maternal chromosomes). These pregnancies occur when a single sperm fertilises an “empty” egg (i.e. an egg containing no DNA) and then duplicates its own chromosomes (rather than joining with the maternal chromosome, as is the case in a normal pregnancy).
Partial hydatidiform moles are known as triploid gestations (i.e. pregnancies that have two paternal and one maternal chromosomes). They occur when two sperm fertilise a single egg. Partial moles sometimes coexist with a foetus. In such instances the foetus will usually have congenital abnormalities and be unviable (i.e. it will not survive). In rare instances a normal foetus will co-occur with a molar pregnancy, in which case there is the possibility of preterm birth.
Hydatidiform moles are benign and confined to the uterine cavity, however in about 10% of cases they develop into malignant tumours. Malignant GTD tumours are either:
- invasive hydatidiform moles: previously benign hydatidiform moles which have transformed;
- choriocarcinoma: a very aggressive form of GTD, which can occur up to 15 years following the previous pregnancy;
- placental site trophoblastic tumours a tumour of the placental site which typically occurs many years after the last pregnancy and is often resistant to chemotherapy; or
- epitheliod trophoblastic tumour: similar to placental site trophoblastic tumours.
These malignant forms of GTD most commonly occur after a previous benign molar pregnancy, however they can also occur after an apparently normal pregnancy (either carried to term or terminated through spontaneous or induced abortion) or an ectopic pregnancy. The extent and sites of metastasis vary depending on the type of tumor:
- Invasive hydatidiform moles typically metastasise locally (i.e. the tumour progressively grows into the uterus walls), however in rare instances metastasis to the vagina, lungs and brain have been documented.
- Choriocarcinomas are a highly malignant form of GTD. They have the ability to enter the bloodstream and metastasise to distant sites, commonly including the vagina, lungs, brain, liver, kidneys and bowel.
Statistics
An estimated 1-3 in 1000 pregnancies are affected by benign hydatidiform moles, however incidence varies internationally and is highest in the Asian region. Some 10% of all hydatidiform moles will become malignant; an estimated 8-15% of complete and 1.5-6% of partial hydatidiform moles.Around 8% of women with a previous molar pregnancy will develop persistent trophoblastic disease (i.e. recurring hydatidiform moles).
In Australia it is estimated that one in 1500 pregnancies is affected by hydatidiform mole.
Risk Factors
All women of reproductive age who have previously been, or are currently pregnant, are at risk of GTD, however women younger than 16 or older than 40 who become pregnant are more likely than women aged 16-39, to develop a molar pregnancy.
There is a one in three chance of GTD in women aged over 50 who become pregnant. Other factors which predispose women to GTD relate to genetics and reproductive history. These include:
- Asian ethnicity: there is a higher incidence of GTD in the Asian region. Women of Asian ethnicity are almost twice as likely to develop GTD than women of non-Asian ethnicity;
- Previous molar pregnancy: around half of all choriocarcinomas appear in women who have previously had GTD and approximately one in seven pregnancies occurring in this group is a molar pregnancy;
- Family history of molar pregnancy: there is evidence that GTD has genetic causes related to mutation of a gene on chromosome 19;
- Dietary Factors: low intake of carotene and animal fat has been associated with increased risk of complete hydatidiform, as has Vitamin A deficiency.
Progression
Benign hydatidiform moles occurs following any type of previous pregnancy, however they most commonly occurs following other molar pregnancies. Scientists are still investigating exactly how placental tissues go on to form moles, however the most likely cause is defective fertilisation, which leads to an abnormal pregnancy, in which the placenta grows, but the embryo does not.
Once formed, a hydatidiform mole can either:
- Spontaneously resolve: that is the mole is expelled from the body via the vagina in circumstances very similar to a miscarriage and no treatment is necessary;
- Develop to invasive hydatidiform moles: that is benign moles may transform to malignant moles and metastasise. The benign tumour may lay dormant for an extended period of time until hormonal changes (often associated with a future pregnancy) stimulate its growth. An estimated 8-15% of complete and 1.5-6% of partial moles take this course; or
- Develop to persistent trophoblastic disease: about 8% of patients will experience recurring hydatidiform moles requiring chemotherapy following surgical treatment.
GTD characterised by malignant trophoblastic tumours is often a result of the transformation of hydatidiform moles, however when it occurs concurrently with a normal pregnancy, there is no clear explanation regarding the cause of the disease. Scientists are still investigating the mechanisms by which benign tumours transform to become malignant.
Symptoms
The most common symptom of molar pregnancy is vaginal bleeding and many women go to the doctor thinking they are having a miscarriage. Other symptoms include abdominal pain and excessive nausea and vomiting (more than normal during early pregnancy). However many women with a molar pregnancy do not display any symptoms.
If the tumour has spread, symptoms include:
- Lung metastasis: may be indicated by a patient coughing up blood or experiencing shortness of breath;
- Brain metastasis: (or metastasis to other central nervous system sites) may indicated dizziness, fainting, fitting or headaches;
- Renal metastasis: may be indicated by dark stools.
Clinical Examination
If a doctor suspects GTD, a physical examination of the pelvic area will usually be performed. An enlarged uterus (i.e. a uterus that is larger than normal for the stage of pregnancy) is the most common physical sign of GTD and this may prompt a doctor to undertake further diagnostic tests.
How is it Diagnosed
A definite diagnosis of GTD can only be made if placental tissues are removed from the woman’s body for examination in a laboratory. As this involves surgery, it is normal for a doctor to perform a number of other tests prior to performing surgery.
Taking measurements of the hormone human Chorionic Gonadotrophin (hCG) (levels of which are elevated during pregnancy, and behave abnormally during a molar pregnancy) can give a doctor a good indication of whether or not GTD is possible.
Transvaginal and transabdominal ultrasound are also commonly used diagnostic tools, which enable doctors to see signs of molar pregnancy (e.g. an abnormal placental mass). The doctor will take an ultrasound of the areas likely to be affected if a molar pregnancy is in fact occurring, and examine the ultrasonic images for evidence of GTD.
Prognosis

Treatment

Treatments for GTD include surgery and chemotherapy, however because chemotherapy has a toxic effect on the body, it is normal for surgical options to be attempted in the first instance. Where surgery is unsuccessful, low-dose single-agent chemotherapy is instituted, followed by high-dose multi-agent chemotherapy in the event of failure.
Surgical treatment
There are two types of surgical techniques which can be used to treat non-metastatic GTD, dilatation and cutterage and hysterectomy.
Dilatation and cutterage is a process of evacuating abnormal tissues from the uterus. It involves a doctor opening the vagina using an instrument known as a speculum, so that the cervix is visible. The cervix is then dilated so that the uterus can be accessed. An instrument called a curette is then used to scrape the walls of the uterus. Following this procedure a doctor will normally measure a patients hormone levels for 8 weeks to a year to ensure that the treatment has been fully successful. If hormone levels rise, further treatment, often with chemotherapy may be needed.
Hysterectomy is sometimes used to remove benign moles when the woman being treated does not wish to have anymore children. Hysterectomy may also be used to treat placental site trophoblast tumours which are often resistant to chemotherapy.
Chemotherapy
A doctor will usually commence chemotherapy as a last resort, due to the toxic effects of chemotherapy medications. However, even as a last resort, chemotherapy will be required in some 8% of patients with benign moles (approximately 15% of complete and 0.5% of partial mole patients). This is when surgery is unsuccessful and the moles return following the surgical treatment.
A doctor will usually assess a patient’s risk of treatment failure, using a system design by the World Health Organisation to assess the risk of treatment failure for GTD. In patients with a low risk score, single agent chemotherapy is usually prescribed. A drug known as Methotrexate is the most commonly prescribed in these circumstances. However there is a risk that patients will become resistant to single agent chemotherapy (i.e. the medication will stop working).
When patients have a high risk score (usually when the tumour has already spread) or when patients become resistant to single-agent chemotherapy, multi-agent chemotherapy is required. This involves taking a combination of different chemotherapy drugs on alternating days. Multi-agent chemotherapy is successful in 80-95% of cases.
Following treatment
Women should not become pregnant again for at least six months after their treatment has been declared successful. The use of oral contraceptives is safe following successful treatment of GTD.
References
- Newlands, E. “Presentation and management of gestational trophoblastic disease and gestational trophoblastic tumours in the United Kingdom” In: Hancock BW, Newlands ES, Berkowitz RS, Cole LA, editors. Gestational Trophoblastic Diseases. 2nd ed. Sheffield: International Society for the Study of Trophoblastic Diseases;2003, pp229-247, available from: www.isstd.org/gtd/ index.html
- Berkowitz RS,Goldstein DP. Chorionic tumours. N Engl J Med 1996;335:1740-8.
- Sebire, N.J. Seckl, M.J. “Gestational Trophoblastic Disease: current management of hidatidiform mole” in BMJ, 2008, 337: 1193-8.
- Lai, C.Y.L. Chan, K.Y.K. Khoo, U. et al “Analysis of gestational trophoblastic disease by genotyping and chromosome in situ hybridization” in Mod Pathol, 2004, 17:40-48.
- Soo-Keat, K. “Clinical aspects of Gestational Trophoblastic Disease: A review based partly on 25 years experience of a statewide registery” in Aust NZ J Obstet Gyneacol, 2003, 43(4): 280-89.
- Fox, H. “Gestational trophoblastic disease” [Editorial] in BMJ, 1997, 314:1363.
- Shih, I. “Gestational trophoblastic neoplasia – pathogenesis and potential therapeutic targets” in Lancet Oncology, 2007, 8:642-50.
- World Health Organisation, International Classification of Disease [Online], 2008, available from: http://www.who.int/classifications/apps/icd/icd10online/
- Sydney Gynaecological Oncology, Gestational Trophoblastic Disease: Fact Sheet, available from: http://www.cs.nsw.gov.au/cancer/sgog/GTD.htm
- Gerulath, A.H. Gestational Trophoblastic Disease: SOCG Clinical Practice Guideline, Society of Obstetricians and Gyneaocologists Canada, available from: www.sogc.org/guidelines
- Tham, B.W.L. Everard, J.E. Tidy, J.A. et al “Gestational Trophoblastic Disease in the Asian population of Morthers in England and North Wales” in BJOG, 2003, 110(6):555-559.
- Sebire, N.J. “Diagnosis of Gestational Trophoblastic Disease in early pregnancy” in Current Diag Pathol, 2003, 8(6)430-440.
- Betel, C Atri, M. Arenson, A. “Sonographic Diagnosis of Gestational Trophoblastic Disease and Comparison with Retained Products of Birth” in J Ultrasound Med, 2006 25:985-993.
- Kohorn, E.I. „The FIGO 2000 staging and risk factor scoring system for gestational trophoblastic neoplasia” In: Hancock BW, Newlands ES, Berkowitz RS, Cole LA, editors. Gestational Trophoblastic Diseases. 2nd ed. Sheffield: International Society for the Study of Trophoblastic Diseases;2003 pp 175-81 available from: www.isstd.org/gtd/ index.html
- Steigrad, S.J. “Epidemiology of gestational trophoblastic diseases” in Best Prac Res Clin Obstet Gynaecol, 2003, 17(6):837-47.
- Khan, F. Everard, J. Ahmed, S. et al, “Low-risk persistent gestational trophoblastic disease treated with low-dose methotrexate: efficacy, acute and long-term effects” in Brit J Cancer, 2003 89:2197-2201.
- Ngan, H.Y.S. Tam, K. Lam, K. Chan, K.K.L. “Methotrexate, Bleomycin and Etoposide in the Treatment of Gestational Trophoblastic Neoplasia” in Obstet Gynecol 2006;107:1012-7.
- Lurain, J.R. “Treatment of Gestational Trophoblastic Tumours” in In: Hancock BW, Newlands ES, Berkowitz RS, Cole LA, editors. Gestational Trophoblastic Diseases. 2nd ed. Sheffield: International Society for the Study of Trophoblastic Diseases;2003, pp 305-18 available from: http://www.isstd.org/gtd/14%20word%20for%20print.pdf
- Consumer Medicine Information – Methotrexate, 2008, Australian Pharmaceutical Board, available from: http://www.pbs.gov.au/html/consumer/home
- McNeish, I.A. Strickland, S. Holden, L. et al “Low-risk persistent gestational trophoblastic disease: outcomes after initial treatment with low-dose methotrexate and folinic acid from 1992-2000” in J Clin Oncol, 2002, 20(7):838-1844.
All content and media on the HealthEngine Blog is created and published online for informational purposes only. It is not intended to be a substitute for professional medical advice and should not be relied on as health or personal advice. Always seek the guidance of your doctor or other qualified health professional with any questions you may have regarding your health or a medical condition. Never disregard the advice of a medical professional, or delay in seeking it because of something you have read on this Website. If you think you may have a medical emergency, call your doctor, go to the nearest hospital emergency department, or call the emergency services immediately.







